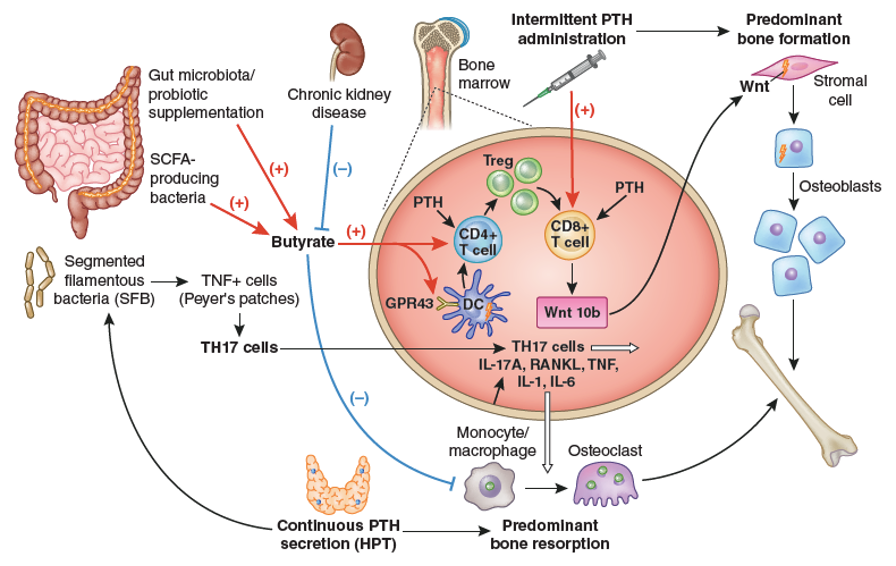
Figure 15. Importance of intestinal microbiota for PTH action in bone. The stimulation of bone anabolism by PTH requires butyrate formation by short chain fatty acid (SCFA) producing gut bacteria. CKD probably reduces its production. Butyrate increases the frequency of regulatory T (Treg) cells in the intestine and in the bone marrow and potentiates the capacity of intermittently administered PTH to induce the differentiation of naïve CD4+ T cells into Tregs, a population of T cells which induces conventional CD8+ T cells to release Wnt10b. This osteogenic Wnt ligand activates Wnt signaling in osteoblastic cells and stimulates bone formation. Butyrate enables intermittent PTH dosing to expand Tregs via GPR43 signaling in dendritic cells (DCs) and GPR43 independent targeting of T cells. Butyrate may also affect bone remodeling by modulating osteoclast genes. The stimulation of bone resorption by PTH requires the presence of segmented filamentous bacteria (SFB) within gut microbiota for the production of Th17 cells in intestinal Peyers’ plaques. Continuously elevated PTH levels lead to TNF+ T cell expansion in the gut and the bone marrow via a microbiota-dependent, but SFB independent mechanism. Furthermore, intestinal TNF producing T cells are required for PTH to increase the number of intestinal Th17 cells, and TNF mediates the migration of intestinal Th17 cells to the bone marrow. This migration depends on upregulation of chemokine receptor CXCR3 and chemokine CCL20. Bone marrow Th17 cells then induce osteoclastogenesis by secreting IL-17A, RANKL, TNF, IL-1, and IL-6. From Massy & Drueke (227).
