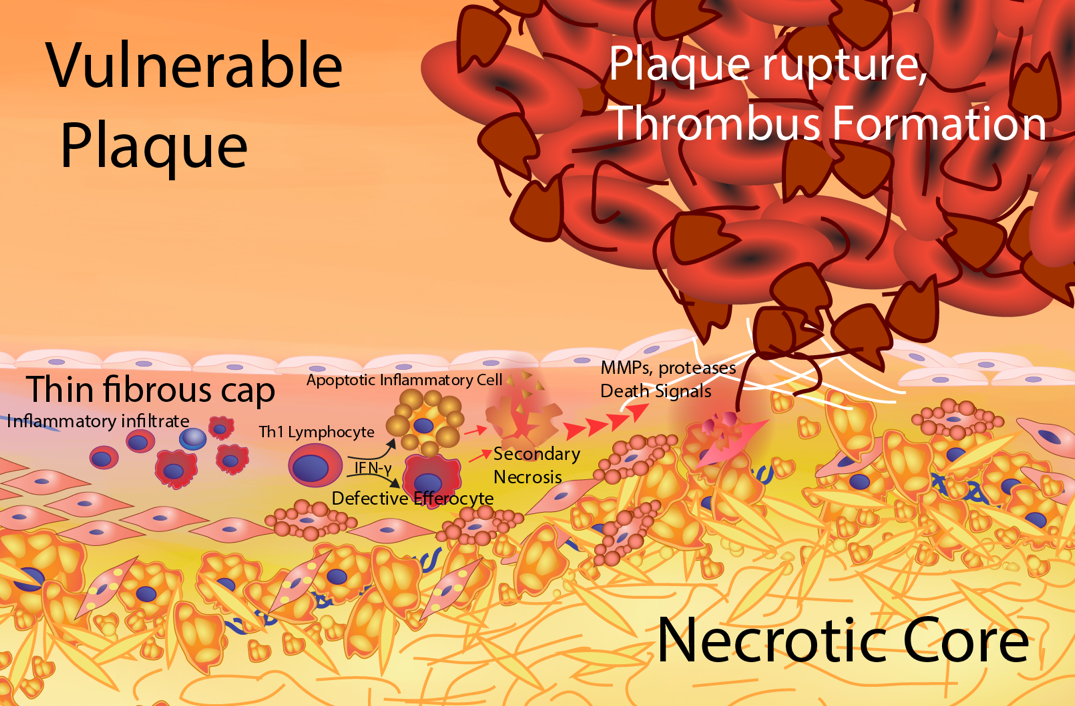
Figure 5. Formation of the vulnerable plaque. The vulnerable plaque results from a heightened, unresolved inflammatory status of the lesion foam cell core. Antigen-specific activation of T helper 1 (Th-1) cells produces interferon gamma (IFNg) resulting in a proinflammatory macrophage phenotype. The proinflammatory macrophage foam cells exhibit enhanced inflammatory cytokine secretion and apoptosis susceptibility. There is less secretion of the anti-inflammatory cytokines, TGF-β and IL-10. In addition, proinflammatory macrophages have impaired atheroprotective functions including cholesterol efflux and efferocytosis. The defective efferocytosis of inflammatory apoptotic macrophages results in secondary necrosis leading to an enlarged necrotic core composed of leaked oxidative and inflammatory components. This unresolved inflammation causes thinning of the fibrous cap resulting from increased smooth muscle cell death, enhanced extracellular matrix degradation and decreased extracellular matrix production. Areas of thin fibrous cap are prone to rupture exposing prothrombotic components to platelets and procoagulation factors leading to thrombus formation and clinical events.
